Phones
Latest about Phones
-
-
 Grab some chipsQualcomm unveils a pair of chips: Snapdragon 6, 4 series to improve the features you actually useBy Nickolas Diaz Published
Grab some chipsQualcomm unveils a pair of chips: Snapdragon 6, 4 series to improve the features you actually useBy Nickolas Diaz Published -
 DealsThe best Verizon deals of May 2026 — FREE Android phones, bundle discounts, and moreBy Patrick Farmer Last updated
DealsThe best Verizon deals of May 2026 — FREE Android phones, bundle discounts, and moreBy Patrick Farmer Last updated -
 That's absurdSkip the upgrade — you can get the 'nearly perfected' Motorola Razr Ultra (2025) for $800 LESS than the 2026 model todayBy Patrick Farmer Published
That's absurdSkip the upgrade — you can get the 'nearly perfected' Motorola Razr Ultra (2025) for $800 LESS than the 2026 model todayBy Patrick Farmer Published -
 Flashy distractionsHonor 600 series hits the UK with big discounts and a pop culture makeoverBy Jay Bonggolto Published
Flashy distractionsHonor 600 series hits the UK with big discounts and a pop culture makeoverBy Jay Bonggolto Published -
 Beta is finally overSamsung’s stable One UI 8.5 update officially lands on older Galaxy phonesBy Jay Bonggolto Published
Beta is finally overSamsung’s stable One UI 8.5 update officially lands on older Galaxy phonesBy Jay Bonggolto Published -
 Software is the spoilerSamsung’s One UI 9 leak just spoiled the Wide Fold’s entire vibeBy Jay Bonggolto Published
Software is the spoilerSamsung’s One UI 9 leak just spoiled the Wide Fold’s entire vibeBy Jay Bonggolto Published -
 Samsung's next big eventSamsung Galaxy Unpacked Summer 2026: How to watch and what to expectBy Sanuj Bhatia Published
Samsung's next big eventSamsung Galaxy Unpacked Summer 2026: How to watch and what to expectBy Sanuj Bhatia Published
-
Explore Phones
Apple iPhone
-
-
 Leadership changeApple's Tim Cook is stepping down as CEO later this year — here's the Apple veteran next upBy Brady Snyder Published
Leadership changeApple's Tim Cook is stepping down as CEO later this year — here's the Apple veteran next upBy Brady Snyder Published -
 How To4 Android phones you should buy instead of the iPhone 17eBy Brady Snyder Published
How To4 Android phones you should buy instead of the iPhone 17eBy Brady Snyder Published -
 iPhone vs. GalaxyiPhone 17e vs. Samsung Galaxy S25 FE: Simplicity or versatility?By Brady Snyder Published
iPhone vs. GalaxyiPhone 17e vs. Samsung Galaxy S25 FE: Simplicity or versatility?By Brady Snyder Published -
 Apple vs GoogleiPhone 17e vs. Google Pixel 10a: Two affordable flagships, one tough decisionBy Sanuj Bhatia Published
Apple vs GoogleiPhone 17e vs. Google Pixel 10a: Two affordable flagships, one tough decisionBy Sanuj Bhatia Published -
 Just as goodNeed to buy an iPhone in 2026? Here's why you should get the iPhone 17By Harish Jonnalagadda Published
Just as goodNeed to buy an iPhone in 2026? Here's why you should get the iPhone 17By Harish Jonnalagadda Published -
 Choices, choicesWith the iPhone 17 selling this well, I don't think Apple cares about low iPhone Air demandBy Brady Snyder Published
Choices, choicesWith the iPhone 17 selling this well, I don't think Apple cares about low iPhone Air demandBy Brady Snyder Published -
 HardwiredThe iPhone Air is the coolest phone I used in 2025By Harish Jonnalagadda Published
HardwiredThe iPhone Air is the coolest phone I used in 2025By Harish Jonnalagadda Published -
 Present or future?I tested the iPhone Air, and it finally lets you choose your compromiseBy Brady Snyder Published
Present or future?I tested the iPhone Air, and it finally lets you choose your compromiseBy Brady Snyder Published -
 Now this is an upgradeI used the iPhone 17 Pro for a week — it is a bigger upgrade than I realizedBy Harish Jonnalagadda Published
Now this is an upgradeI used the iPhone 17 Pro for a week — it is a bigger upgrade than I realizedBy Harish Jonnalagadda Published
-
Asus
-
-
 Bye for now, ASUSI'm going to miss new ASUS phones in 2026 for this one reasonBy Brady Snyder Published
Bye for now, ASUSI'm going to miss new ASUS phones in 2026 for this one reasonBy Brady Snyder Published -
 Finally!Asus ROG Phone 9 and Zenfone 12 Ultra users can now upgrade to Android 16By Brady Snyder Published
Finally!Asus ROG Phone 9 and Zenfone 12 Ultra users can now upgrade to Android 16By Brady Snyder Published -
 Game onASUS ROG Phone 9 Pro review: Ultimate bragging rightsBy Harish Jonnalagadda Last updated
Game onASUS ROG Phone 9 Pro review: Ultimate bragging rightsBy Harish Jonnalagadda Last updated -
 ContradictionsASUS Zenfone 12 Ultra review: I really like this phone, but you shouldn't buy itBy Harish Jonnalagadda Published
ContradictionsASUS Zenfone 12 Ultra review: I really like this phone, but you shouldn't buy itBy Harish Jonnalagadda Published -
 Zen-aestheticASUS reveals the Zenfone 12 Ultra with a spotless design and a powerful flagship chipBy Nickolas Diaz Published
Zen-aestheticASUS reveals the Zenfone 12 Ultra with a spotless design and a powerful flagship chipBy Nickolas Diaz Published -
 A little cheaperThe budget ASUS ROG Phone 9 FE is finally here with a 2023 chip and gaming AIBy Nickolas Diaz Published
A little cheaperThe budget ASUS ROG Phone 9 FE is finally here with a 2023 chip and gaming AIBy Nickolas Diaz Published -
 For the hopefulThe ASUS ROG Phone 9 'FE' is rumored to have a launch on the horizonBy Nickolas Diaz Published
For the hopefulThe ASUS ROG Phone 9 'FE' is rumored to have a launch on the horizonBy Nickolas Diaz Published -
 Ultimate ZenASUS Zenfone 11 Ultra review: Identity crisisBy Harish Jonnalagadda Published
Ultimate ZenASUS Zenfone 11 Ultra review: Identity crisisBy Harish Jonnalagadda Published -
 Asus ROG adds a new ultra-fast Wi-Fi 6E gaming router to its lineupBy Samuel Contreras Published
Asus ROG adds a new ultra-fast Wi-Fi 6E gaming router to its lineupBy Samuel Contreras Published
-
Carriers
-
-
 DealsThe best Verizon deals of May 2026 — FREE Android phones, bundle discounts, and moreBy Patrick Farmer Last updated
DealsThe best Verizon deals of May 2026 — FREE Android phones, bundle discounts, and moreBy Patrick Farmer Last updated -
 Join the partyThe best T-Mobile deals of May 2026: Galaxy S26 deals, home internet savings, and moreBy Patrick Farmer Last updated
Join the partyThe best T-Mobile deals of May 2026: Galaxy S26 deals, home internet savings, and moreBy Patrick Farmer Last updated -
 Free gift card alertA new Verizon deal apparently just gives you $100 to try the Samsung Galaxy S26 — here's how it worksBy Patrick Farmer Published
Free gift card alertA new Verizon deal apparently just gives you $100 to try the Samsung Galaxy S26 — here's how it worksBy Patrick Farmer Published -
 Amazing savingsVerizon brings back bundle deal that gets you a free Samsung Galaxy S26 Plus, Galaxy Watch 8, AND $100 gift card - so what's the catch?By Patrick Farmer Published
Amazing savingsVerizon brings back bundle deal that gets you a free Samsung Galaxy S26 Plus, Galaxy Watch 8, AND $100 gift card - so what's the catch?By Patrick Farmer Published -
 5G for cheapMetro by T-Mobile is slashing 50% off its unlimited plan — score six months for only $20 per month!By Patrick Farmer Published
5G for cheapMetro by T-Mobile is slashing 50% off its unlimited plan — score six months for only $20 per month!By Patrick Farmer Published -
 Easy on the eyesThis is not a test: T-Mobile will already give you the TCL NXTPAPER 70 Pro for FREE with new dealBy Patrick Farmer Published
Easy on the eyesThis is not a test: T-Mobile will already give you the TCL NXTPAPER 70 Pro for FREE with new dealBy Patrick Farmer Published -
 Minty savings!The best Mint Mobile deals of April 2026 — 50% off wireless, 5G home internet discounts, and moreBy Patrick Farmer Last updated
Minty savings!The best Mint Mobile deals of April 2026 — 50% off wireless, 5G home internet discounts, and moreBy Patrick Farmer Last updated -
 Just keeps getting betterMint Mobile is now carving up to $900 OFF the Galaxy S26 — here is why it's one of the best Samsung deals on the web todayBy Patrick Farmer Published
Just keeps getting betterMint Mobile is now carving up to $900 OFF the Galaxy S26 — here is why it's one of the best Samsung deals on the web todayBy Patrick Farmer Published -
 Travel smart!Best wireless plans for international travel 2026By Patrick Farmer Last updated
Travel smart!Best wireless plans for international travel 2026By Patrick Farmer Last updated
-
Google Fi
-
-
 It's not just youIf your Google Fi says your SIM isn't active, here's what's going onBy Sanuj Bhatia Published
It's not just youIf your Google Fi says your SIM isn't active, here's what's going onBy Sanuj Bhatia Published -
 Lots of choices!The best Google Fi Wireless alternatives in 2026By Samuel Contreras Last updated
Lots of choices!The best Google Fi Wireless alternatives in 2026By Samuel Contreras Last updated -
 Save with Fi!Google Fi Wireless plans, deals, and prices: everything you need to knowBy Samuel Contreras Last updated
Save with Fi!Google Fi Wireless plans, deals, and prices: everything you need to knowBy Samuel Contreras Last updated -
 AI power-upGoogle Fi is using AI to make your phone service less annoyingBy Jay Bonggolto Published
AI power-upGoogle Fi is using AI to make your phone service less annoyingBy Jay Bonggolto Published -
 Sharing is caring!Which MVNOs allow tethering?By Samuel Contreras Last updated
Sharing is caring!Which MVNOs allow tethering?By Samuel Contreras Last updated -
 Tenth anniversaryGoogle Fi celebrates 10 years with a new affordable plan to take on T-MobileBy Vishnu Sarangapurkar Published
Tenth anniversaryGoogle Fi celebrates 10 years with a new affordable plan to take on T-MobileBy Vishnu Sarangapurkar Published -
 Ask JerryAsk Jerry: Will my phone work outside of the US?By Jerry Hildenbrand Published
Ask JerryAsk Jerry: Will my phone work outside of the US?By Jerry Hildenbrand Published -
 Pixel 8a spottedThe Pixel 8a may have just appeared in a Google Fi Wireless adBy Vishnu Sarangapurkar Published
Pixel 8a spottedThe Pixel 8a may have just appeared in a Google Fi Wireless adBy Vishnu Sarangapurkar Published -
 Two Great Options!Tello vs. Google Fi: Which flexible prepaid carrier is the best?By Samuel Contreras Last updated
Two Great Options!Tello vs. Google Fi: Which flexible prepaid carrier is the best?By Samuel Contreras Last updated
-
Google Pixel
-
-
 Rolling out fixesGoogle's May security patch for Pixels is here, targeting charging and displayBy Nickolas Diaz Published
Rolling out fixesGoogle's May security patch for Pixels is here, targeting charging and displayBy Nickolas Diaz Published -
 Mega savingsForget the Pixel 10a — Mint Mobile will give you a base Google Pixel 10 AND a year of Unlimited for only $480By Patrick Farmer Published
Mega savingsForget the Pixel 10a — Mint Mobile will give you a base Google Pixel 10 AND a year of Unlimited for only $480By Patrick Farmer Published -
 Interesting so farThese Pixel 11 series leaks are huge, and so is this 'Pixel Glow' design rumorBy Nickolas Diaz Published
Interesting so farThese Pixel 11 series leaks are huge, and so is this 'Pixel Glow' design rumorBy Nickolas Diaz Published -
 Hoped for moreGoogle's Tensor G6 rumors nearly had me hyped, but it might let me downBy Nickolas Diaz Published
Hoped for moreGoogle's Tensor G6 rumors nearly had me hyped, but it might let me downBy Nickolas Diaz Published -
 It's glowingMore 'Pixel Glow' rumors light up Google's plans, teases its AI's involvementBy Nickolas Diaz Published
It's glowingMore 'Pixel Glow' rumors light up Google's plans, teases its AI's involvementBy Nickolas Diaz Published -
 AI fuels growthAI in Google Search is paying off: Alphabet posts strong Q1 2026 growthBy Sanuj Bhatia Published
AI fuels growthAI in Google Search is paying off: Alphabet posts strong Q1 2026 growthBy Sanuj Bhatia Published -
 Hidden in codeGoogle's Android 17 preview may have spilled Pixel 11 Pro Fold detailsBy Sanuj Bhatia Published
Hidden in codeGoogle's Android 17 preview may have spilled Pixel 11 Pro Fold detailsBy Sanuj Bhatia Published -
 Pixel Price DropThe Google Pixel 10 just slid back to its lowest price EVER with 31% off at AmazonBy Zachary David Published
Pixel Price DropThe Google Pixel 10 just slid back to its lowest price EVER with 31% off at AmazonBy Zachary David Published -
 Pixel bugPixel phones are seeing unusual battery drain after the latest updateBy Sanuj Bhatia Published
Pixel bugPixel phones are seeing unusual battery drain after the latest updateBy Sanuj Bhatia Published
-
Honor
-
-
 Flashy distractionsHonor 600 series hits the UK with big discounts and a pop culture makeoverBy Jay Bonggolto Published
Flashy distractionsHonor 600 series hits the UK with big discounts and a pop culture makeoverBy Jay Bonggolto Published -
 It's magicHonor Connect, photography updates carry the Magic 8 Pro's huge April patchBy Nickolas Diaz Published
It's magicHonor Connect, photography updates carry the Magic 8 Pro's huge April patchBy Nickolas Diaz Published -
 Good stuffHonor 600 Pro review: This phone is so good that it surprised meBy Nirave Gondhia Published
Good stuffHonor 600 Pro review: This phone is so good that it surprised meBy Nirave Gondhia Published -
 New Honor phoneThe new Honor 600 series brings flagship cameras and upgraded AI featuresBy Nirave Gondhia Published
New Honor phoneThe new Honor 600 series brings flagship cameras and upgraded AI featuresBy Nirave Gondhia Published -
 What an honorHonor 600 review: Flagship feels, mid-range priceBy Nicholas Sutrich Published
What an honorHonor 600 review: Flagship feels, mid-range priceBy Nicholas Sutrich Published -
 The thin phone of your dreamsThe Magic 8 Pro Air proves that Apple and Samsung got their thin phones wrongBy Nirave Gondhia Published
The thin phone of your dreamsThe Magic 8 Pro Air proves that Apple and Samsung got their thin phones wrongBy Nirave Gondhia Published -
 Android Central LabsThe Honor MagicPad 4's display does something I've never seen before, but there's one annoying catchBy Nicholas Sutrich Published
Android Central LabsThe Honor MagicPad 4's display does something I've never seen before, but there's one annoying catchBy Nicholas Sutrich Published -
 Weird, in a good wayHonor's Robot Phone is one of the strangest (and coolest) things I've seen at MWC 2026By Sanuj Bhatia Published
Weird, in a good wayHonor's Robot Phone is one of the strangest (and coolest) things I've seen at MWC 2026By Sanuj Bhatia Published -
 MWC excitementHonor has the courage to do what Samsung won't, and that's convinced me of the Magic V6's greatnessBy Nicholas Sutrich Published
MWC excitementHonor has the courage to do what Samsung won't, and that's convinced me of the Magic V6's greatnessBy Nicholas Sutrich Published
-
Huawei
-
-
 Wide is the new tallThe wide foldable is finally here, and it didn’t come from SamsungBy Jay Bonggolto Published
Wide is the new tallThe wide foldable is finally here, and it didn’t come from SamsungBy Jay Bonggolto Published -
 Soaring aboveI used Huawei's Mate X7 to take photos of the world's tallest building, and I'm convinced that this foldable has the best camerasBy Harish Jonnalagadda Published
Soaring aboveI used Huawei's Mate X7 to take photos of the world's tallest building, and I'm convinced that this foldable has the best camerasBy Harish Jonnalagadda Published -
 Coming BackHuawei's alleged Mate XT sequel might have some spicy upgrades up its sleeveBy Nickolas Diaz Published
Coming BackHuawei's alleged Mate XT sequel might have some spicy upgrades up its sleeveBy Nickolas Diaz Published -
 What could it be?Jackie Chan was spotted using a foldable on TikTok, and we've got our top suspectsBy Nandika Ravi Published
What could it be?Jackie Chan was spotted using a foldable on TikTok, and we've got our top suspectsBy Nandika Ravi Published -
 The bestHuawei Mate XT camera review: Putting all other foldables to shameBy Harish Jonnalagadda Published
The bestHuawei Mate XT camera review: Putting all other foldables to shameBy Harish Jonnalagadda Published -
 Why not both?Huawei's new flip phone blurs the line between foldablesBy Nickolas Diaz Published
Why not both?Huawei's new flip phone blurs the line between foldablesBy Nickolas Diaz Published -
 Leveling upHuawei's tri-fold Mate XT is the most exciting phone I've used in a long timeBy Harish Jonnalagadda Published
Leveling upHuawei's tri-fold Mate XT is the most exciting phone I've used in a long timeBy Harish Jonnalagadda Published -
 Best Huawei phones 2022By Harish Jonnalagadda Last updated
Best Huawei phones 2022By Harish Jonnalagadda Last updated -
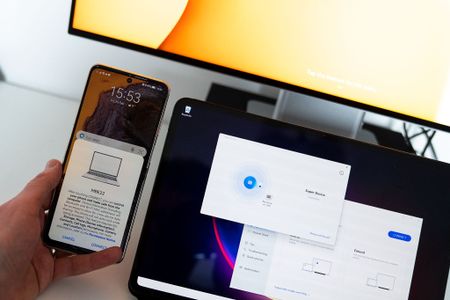 Huawei's 'Super Device' is greater than the sum of its partsBy Alex Dobie Published
Huawei's 'Super Device' is greater than the sum of its partsBy Alex Dobie Published
-
MediaTek
-
-
 Powered-upMediaTek's Dimensity 9500s, 8500 surge with power for even more Android phonesBy Nickolas Diaz Published
Powered-upMediaTek's Dimensity 9500s, 8500 surge with power for even more Android phonesBy Nickolas Diaz Published -
 OVER 9000MediaTek's new powerhouse chip is the unicorn you'll never get to seeBy Jerry Hildenbrand Published
OVER 9000MediaTek's new powerhouse chip is the unicorn you'll never get to seeBy Jerry Hildenbrand Published -
 Flagship powerThe MediaTek Dimensity 9500 is the Android powerhouse you'd expectBy Michael L Hicks Published
Flagship powerThe MediaTek Dimensity 9500 is the Android powerhouse you'd expectBy Michael L Hicks Published -
 Chip launch confirmedMediaTek confirms the date for its next flagship chip launch, and the timing is interestingBy Sanuj Bhatia Published
Chip launch confirmedMediaTek confirms the date for its next flagship chip launch, and the timing is interestingBy Sanuj Bhatia Published -
 Early DaysVery early MediaTek Dimensity 9500 benchmark leaves us waiting for moreBy Nickolas Diaz Published
Early DaysVery early MediaTek Dimensity 9500 benchmark leaves us waiting for moreBy Nickolas Diaz Published -
 Agentic AIMediaTek highlights Dimensity 9400 Plus with edge-AI specs and 20% boost in Gen AIBy Nickolas Diaz Published
Agentic AIMediaTek highlights Dimensity 9400 Plus with edge-AI specs and 20% boost in Gen AIBy Nickolas Diaz Published -
 Next-gen processorMediaTek's Kompanio Ultra will 'redefine' Chromebook Plus performance and AIBy Nickolas Diaz Published
Next-gen processorMediaTek's Kompanio Ultra will 'redefine' Chromebook Plus performance and AIBy Nickolas Diaz Published -
 MediaTek unveils Dimensity 700 for budget 5G phones and a 6nm Chromebook chipsetBy Harish Jonnalagadda Published
MediaTek unveils Dimensity 700 for budget 5G phones and a 6nm Chromebook chipsetBy Harish Jonnalagadda Published
-
Motorola
-
-
 That's absurdSkip the upgrade — you can get the 'nearly perfected' Motorola Razr Ultra (2025) for $800 LESS than the 2026 model todayBy Patrick Farmer Published
That's absurdSkip the upgrade — you can get the 'nearly perfected' Motorola Razr Ultra (2025) for $800 LESS than the 2026 model todayBy Patrick Farmer Published -
 Juice 'em up!How fast is the Motorola Razr 2026 charging speed?By Rajat Sharma Published
Juice 'em up!How fast is the Motorola Razr 2026 charging speed?By Rajat Sharma Published -
 Worth it?Motorola Razr Ultra 2026 vs. Razr Ultra 2025: Spot the differenceBy Roydon Cerejo Published
Worth it?Motorola Razr Ultra 2026 vs. Razr Ultra 2025: Spot the differenceBy Roydon Cerejo Published -
 Flipping Good DealsThe budget-friendly Motorola Razr (2025) just got budget-friendlier with a huge $150 discount at Best BuyBy Zachary David Published
Flipping Good DealsThe budget-friendly Motorola Razr (2025) just got budget-friendlier with a huge $150 discount at Best BuyBy Zachary David Published -
 Stylish yet durable!Is the Motorola Razr 2026 waterproof?By Rajat Sharma Published
Stylish yet durable!Is the Motorola Razr 2026 waterproof?By Rajat Sharma Published -
 Fighting chanceMotorola Razr Fold vs. Google Pixel 10 Pro Fold: A new competitor unfoldsBy Roydon Cerejo Last updated
Fighting chanceMotorola Razr Fold vs. Google Pixel 10 Pro Fold: A new competitor unfoldsBy Roydon Cerejo Last updated -
 AC PollsThe Motorola Razr 2026 series is here, and preorders begin soon. Which model would you buy?By Derrek Lee Published
AC PollsThe Motorola Razr 2026 series is here, and preorders begin soon. Which model would you buy?By Derrek Lee Published -
 Editor's Desk6 reasons you should buy a 2026 Motorola Razr flip phone, and 4 reasons you should skip themBy Derrek Lee Published
Editor's Desk6 reasons you should buy a 2026 Motorola Razr flip phone, and 4 reasons you should skip themBy Derrek Lee Published -
 Razr edgeMotorola Razr 2027: What we want to seeBy Harish Jonnalagadda Published
Razr edgeMotorola Razr 2027: What we want to seeBy Harish Jonnalagadda Published
-
Nothing
-
-
 AC HacksUnlock the hidden potential of your Nothing Phone 4a Pro with these 4 Essential tips and tricksBy Nicholas Sutrich Published
AC HacksUnlock the hidden potential of your Nothing Phone 4a Pro with these 4 Essential tips and tricksBy Nicholas Sutrich Published -
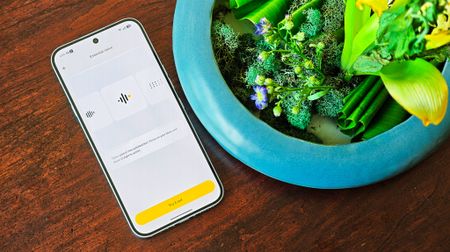 Android Central LabsNothing's Essential Voice redefined how I talk to my phone, and it's about time someone fixed this problemBy Nicholas Sutrich Published
Android Central LabsNothing's Essential Voice redefined how I talk to my phone, and it's about time someone fixed this problemBy Nicholas Sutrich Published -
 Your voiceNothing's giving its phones an Essential Voice to usher in smarter transcriptionsBy Nickolas Diaz Published
Your voiceNothing's giving its phones an Essential Voice to usher in smarter transcriptionsBy Nickolas Diaz Published -
 The SIM tool trapThe Nothing Phone 4a Pro is nearly unbreakable, except for one design flawBy Jay Bonggolto Published
The SIM tool trapThe Nothing Phone 4a Pro is nearly unbreakable, except for one design flawBy Jay Bonggolto Published -
 Not AirDropNothing's new tool makes sharing between Android and desktop easier — and it does one thing differently [Update]By Sanuj Bhatia Published
Not AirDropNothing's new tool makes sharing between Android and desktop easier — and it does one thing differently [Update]By Sanuj Bhatia Published -
 Nothing vs GoogleNothing Phone 4a Pro vs. Google Pixel 10a: A style-first phone vs a smarter oneBy Sanuj Bhatia Published
Nothing vs GoogleNothing Phone 4a Pro vs. Google Pixel 10a: A style-first phone vs a smarter oneBy Sanuj Bhatia Published -
 Nothing beats a PixelNothing Phone 4a Pro review: The right phone at the right timeBy Nicholas Sutrich Published
Nothing beats a PixelNothing Phone 4a Pro review: The right phone at the right timeBy Nicholas Sutrich Published -
 Nothing reportedly interested in AI smart glasses that might be a year awayBy Nickolas Diaz Published
Nothing reportedly interested in AI smart glasses that might be a year awayBy Nickolas Diaz Published -
 Save 25%This stylish Android phone from Nothing has a rear display and is $200 OFFBy Brady Snyder Published
Save 25%This stylish Android phone from Nothing has a rear display and is $200 OFFBy Brady Snyder Published
-
Oneplus
-
-
 Merged?Major reports say OnePlus, Realme merge, but there's a lot still in the darkBy Nickolas Diaz Published
Merged?Major reports say OnePlus, Realme merge, but there's a lot still in the darkBy Nickolas Diaz Published -
 Press StartYeah, the OnePlus Ace 6 Ultra is a phone, but it's mimicking handheld consolesBy Nickolas Diaz Published
Press StartYeah, the OnePlus Ace 6 Ultra is a phone, but it's mimicking handheld consolesBy Nickolas Diaz Published -
 Not looking goodOnePlus' Europe exit isn't official yet, but the signs aren't greatBy Sanuj Bhatia Published
Not looking goodOnePlus' Europe exit isn't official yet, but the signs aren't greatBy Sanuj Bhatia Published -
 OnePlus Nord 6 review: The endurance champion of 2026By Harish Jonnalagadda Published
OnePlus Nord 6 review: The endurance champion of 2026By Harish Jonnalagadda Published -
 Never settleBest OnePlus phones 2026By Harish Jonnalagadda Last updated
Never settleBest OnePlus phones 2026By Harish Jonnalagadda Last updated -
 A Mid-Range FlagshipThe OnePlus 15R just got a rare discount for the Big Spring Sale, but is the phone actually worth it?By Zachary David Published
A Mid-Range FlagshipThe OnePlus 15R just got a rare discount for the Big Spring Sale, but is the phone actually worth it?By Zachary David Published -
 Better value hereWhy pay for the Galaxy S26 Ultra? This phone makes way more sense — and it just received a historic discount for Amazon's Big Spring SaleBy Sanuj Bhatia Published
Better value hereWhy pay for the Galaxy S26 Ultra? This phone makes way more sense — and it just received a historic discount for Amazon's Big Spring SaleBy Sanuj Bhatia Published -
 Leadership exitOnePlus executive who dismissed shutdown rumors has now stepped downBy Sanuj Bhatia Published
Leadership exitOnePlus executive who dismissed shutdown rumors has now stepped downBy Sanuj Bhatia Published -
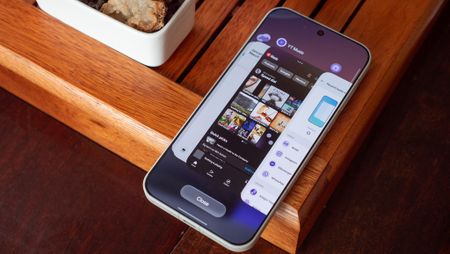 How ToOnePlus ruined its task switcher UI, but there's a secret way to get the good one backBy Nicholas Sutrich Published
How ToOnePlus ruined its task switcher UI, but there's a secret way to get the good one backBy Nicholas Sutrich Published
-
Oppo
-
-
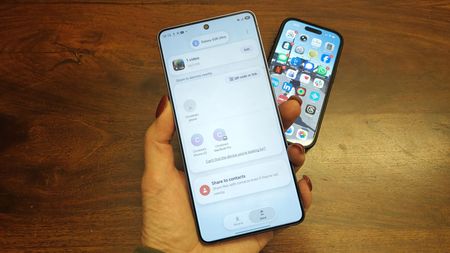 Finally expandingAirDrop sharing appears to be expanding beyond Pixel and Galaxy phonesBy Sanuj Bhatia Published
Finally expandingAirDrop sharing appears to be expanding beyond Pixel and Galaxy phonesBy Sanuj Bhatia Published -
 Sort it outI finally got a handle on my expenses thanks to this AI feature on the Find X9 UltraBy Harish Jonnalagadda Published
Sort it outI finally got a handle on my expenses thanks to this AI feature on the Find X9 UltraBy Harish Jonnalagadda Published -
 OPPO Find X9 Ultra review: This world-class camera is also a phoneBy Harish Jonnalagadda Published
OPPO Find X9 Ultra review: This world-class camera is also a phoneBy Harish Jonnalagadda Published -
 OPPO Find X9 Ultra hands-on: Is this the best camera phone of 2026?By Harish Jonnalagadda Published
OPPO Find X9 Ultra hands-on: Is this the best camera phone of 2026?By Harish Jonnalagadda Published -
 Just look at itOPPO's Reno 15 Pro and Reno 15 are the flagship killers you deserve in 2026By Harish Jonnalagadda Published
Just look at itOPPO's Reno 15 Pro and Reno 15 are the flagship killers you deserve in 2026By Harish Jonnalagadda Published -
 Finding OPPOOPPO Find N6 vs. Samsung Galaxy Z Fold 7: We have a clear winnerBy Roydon Cerejo Published
Finding OPPOOPPO Find N6 vs. Samsung Galaxy Z Fold 7: We have a clear winnerBy Roydon Cerejo Published -
 Ultra worldwideOPPO puts the Find X9 Ultra and 'global' in the same sentence, teases April debutBy Nickolas Diaz Published
Ultra worldwideOPPO puts the Find X9 Ultra and 'global' in the same sentence, teases April debutBy Nickolas Diaz Published -
 How ToOPPO's Find N6 foldable has a unique multitasking feature that Google needs to steal immediatelyBy Harish Jonnalagadda Published
How ToOPPO's Find N6 foldable has a unique multitasking feature that Google needs to steal immediatelyBy Harish Jonnalagadda Published -
 Finding greatnessOPPO Find N6 review: The next step in foldable evolutionBy Harish Jonnalagadda Published
Finding greatnessOPPO Find N6 review: The next step in foldable evolutionBy Harish Jonnalagadda Published
-
Qualcomm
-
-
 Grab some chipsQualcomm unveils a pair of chips: Snapdragon 6, 4 series to improve the features you actually useBy Nickolas Diaz Published
Grab some chipsQualcomm unveils a pair of chips: Snapdragon 6, 4 series to improve the features you actually useBy Nickolas Diaz Published -
 Qualcomm FY Q2 2026 earnings highlight a mobile industry under pressure from the memory shortageBy Derrek Lee Published
Qualcomm FY Q2 2026 earnings highlight a mobile industry under pressure from the memory shortageBy Derrek Lee Published -
 Elite, Pro, hello!Snapdragon 8 Elite Gen 6 CPU rumors join the 'powerful' 6 Pro in this early reportBy Nickolas Diaz Published
Elite, Pro, hello!Snapdragon 8 Elite Gen 6 CPU rumors join the 'powerful' 6 Pro in this early reportBy Nickolas Diaz Published -
 How To4 ways Snapdragon Wear Elite will make your next wearable more usefulBy Brady Snyder Published
How To4 ways Snapdragon Wear Elite will make your next wearable more usefulBy Brady Snyder Published -
 Smarter watches aheadQualcomm's new Snapdragon Wear Elite wants to make AI wearables actually smartBy Sanuj Bhatia Published
Smarter watches aheadQualcomm's new Snapdragon Wear Elite wants to make AI wearables actually smartBy Sanuj Bhatia Published -
 all-in-one connectivity chipQualcomm's Wi-Fi 8 chips are here, and they actually fix the stuff that annoys youBy Jay Bonggolto Published
all-in-one connectivity chipQualcomm's Wi-Fi 8 chips are here, and they actually fix the stuff that annoys youBy Jay Bonggolto Published -
 Faster speedsQualcomm's X105 modem will keep you connected in elevators and parking garagesBy Brady Snyder Published
Faster speedsQualcomm's X105 modem will keep you connected in elevators and parking garagesBy Brady Snyder Published -
 EarningsQualcomm reports record earnings, but things aren't looking goodBy Derrek Lee Published
EarningsQualcomm reports record earnings, but things aren't looking goodBy Derrek Lee Published -
 Elite... is also Pro?Qualcomm's next 'Elite' Snapdragon could go Pro for this year's flagshipsBy Nickolas Diaz Published
Elite... is also Pro?Qualcomm's next 'Elite' Snapdragon could go Pro for this year's flagshipsBy Nickolas Diaz Published
-
RedMagic
-
-
 fresh faceRedMagic 11 Air Trace Edition is the same great gaming phone in a louder shirtBy Jay Bonggolto Published
fresh faceRedMagic 11 Air Trace Edition is the same great gaming phone in a louder shirtBy Jay Bonggolto Published -
 How ToThese four features make the Redmagic 11 Air a beast of a gaming phoneBy Harish Jonnalagadda Published
How ToThese four features make the Redmagic 11 Air a beast of a gaming phoneBy Harish Jonnalagadda Published -
 Slimmer, not weakerThe RedMagic 11 Air is surprisingly thin, and I’m obsessed with its massive 7,000mAh batteryBy Jay Bonggolto Published
Slimmer, not weakerThe RedMagic 11 Air is surprisingly thin, and I’m obsessed with its massive 7,000mAh batteryBy Jay Bonggolto Published -
 What're we playing?RedMagic 11 Air is a cool, crisp mobile gaming guru with the series' biggest 7,000mAh batteryBy Nickolas Diaz Published
What're we playing?RedMagic 11 Air is a cool, crisp mobile gaming guru with the series' biggest 7,000mAh batteryBy Nickolas Diaz Published -
 Cooling ReimaginedRedMagic just dropped a gaming phone with a literal water cooler insideBy Jay Bonggolto Published
Cooling ReimaginedRedMagic just dropped a gaming phone with a literal water cooler insideBy Jay Bonggolto Published -
 Stylish powerRedmagic 10S Pro review: Beating ASUS at its own gameBy Harish Jonnalagadda Published
Stylish powerRedmagic 10S Pro review: Beating ASUS at its own gameBy Harish Jonnalagadda Published -
 Game TimeThere may be a REDMAGIC gaming tablet on the horizon with a smaller screenBy Nickolas Diaz Published
Game TimeThere may be a REDMAGIC gaming tablet on the horizon with a smaller screenBy Nickolas Diaz Published -
 sleek and powerfulRedMagic's latest gaming phone is smart and powerful without the bulkBy Jay Bonggolto Published
sleek and powerfulRedMagic's latest gaming phone is smart and powerful without the bulkBy Jay Bonggolto Published -
 Game onThe RedMagic 10 Pro is a mobile gaming BEAST, and it just scored its first major discount at AmazonBy Patrick Farmer Published
Game onThe RedMagic 10 Pro is a mobile gaming BEAST, and it just scored its first major discount at AmazonBy Patrick Farmer Published
-
Samsung Galaxy
-
-
 Beta is finally overSamsung’s stable One UI 8.5 update officially lands on older Galaxy phonesBy Jay Bonggolto Published
Beta is finally overSamsung’s stable One UI 8.5 update officially lands on older Galaxy phonesBy Jay Bonggolto Published -
 Software is the spoilerSamsung’s One UI 9 leak just spoiled the Wide Fold’s entire vibeBy Jay Bonggolto Published
Software is the spoilerSamsung’s One UI 9 leak just spoiled the Wide Fold’s entire vibeBy Jay Bonggolto Published -
 Samsung's next big eventSamsung Galaxy Unpacked Summer 2026: How to watch and what to expectBy Sanuj Bhatia Published
Samsung's next big eventSamsung Galaxy Unpacked Summer 2026: How to watch and what to expectBy Sanuj Bhatia Published -
 Samsung vs MotorolaSamsung Galaxy Z Flip 8 vs. Motorola Razr Ultra 2026: The fight for the best flip phone of the yearBy Sanuj Bhatia Published
Samsung vs MotorolaSamsung Galaxy Z Flip 8 vs. Motorola Razr Ultra 2026: The fight for the best flip phone of the yearBy Sanuj Bhatia Published -
 Samsung vs MotorolaSamsung Galaxy Z Fold 8 vs. Motorola Razr Fold: Samsung's next foldable has serious competitionBy Sanuj Bhatia Published
Samsung vs MotorolaSamsung Galaxy Z Fold 8 vs. Motorola Razr Fold: Samsung's next foldable has serious competitionBy Sanuj Bhatia Published -
 Three for free?This viral Verizon deal supposedly gets you a free Samsung phone, tablet, AND smartwatch — but how exactly does it work?By Patrick Farmer Published
Three for free?This viral Verizon deal supposedly gets you a free Samsung phone, tablet, AND smartwatch — but how exactly does it work?By Patrick Farmer Published -
 Videos take overSamsung News just got much better with new video stories and top sourcesBy Sanuj Bhatia Published
Videos take overSamsung News just got much better with new video stories and top sourcesBy Sanuj Bhatia Published -
 the wait is over?Galaxy S25 owners could see stable One UI 8.5 land in just a few daysBy Jay Bonggolto Published
the wait is over?Galaxy S25 owners could see stable One UI 8.5 land in just a few daysBy Jay Bonggolto Published -
 Worthwhile?This one rumored Galaxy Z Flip 8 display upgrade might've brought my interest backBy Nickolas Diaz Published
Worthwhile?This one rumored Galaxy Z Flip 8 display upgrade might've brought my interest backBy Nickolas Diaz Published
-
More about Phones
-
-
 Samsung's next big eventSamsung Galaxy Unpacked Summer 2026: How to watch and what to expectBy Sanuj Bhatia Published
Samsung's next big eventSamsung Galaxy Unpacked Summer 2026: How to watch and what to expectBy Sanuj Bhatia Published -
 Juice 'em up!How fast is the Motorola Razr 2026 charging speed?By Rajat Sharma Published
Juice 'em up!How fast is the Motorola Razr 2026 charging speed?By Rajat Sharma Published -
 Worth it?Motorola Razr Ultra 2026 vs. Razr Ultra 2025: Spot the differenceBy Roydon Cerejo Published
Worth it?Motorola Razr Ultra 2026 vs. Razr Ultra 2025: Spot the differenceBy Roydon Cerejo Published
-