What makes a phone battery explode?

Get the latest news from Android Central, your trusted companion in the world of Android
You are now subscribed
Your newsletter sign-up was successful
Exploding battery talk is something you can't escape right now, thanks to a problem with the Samsung Galaxy Note 7. It's good discussion to have: the more people talk about it, the less likely it is for a child to get hurt from a battery failure. As much as we hate to see a product we like experience safety issues, we hate seeing people get hurt far more.
But the Note 7 isn't the first phone to ever go through battery problems, and it won't be the last. There will always be isolated incidents of batteries failing as long as we use phones with lithium-ion cells, and the Note 7 isn't the first phone whose battery needed a widespread recall because something is wrong under the hood — as long-time Nokia fans know too well. It happens. It's never a good thing, but it's a thing. Let's talk about why it can happen.
How your phone battery works

The first thing we need to understand is how exactly the lithium-ion battery in your phone works. The name gives us a hint — electricity is carried from one electrode to another using charged lithium ions.
Article continues belowElectricity meets chemistry meets metallurgy — that's what makes a battery possible.
Lithium-ion batteries store, transfer and release energy because of natural chemical reactions. The battery has two electrodes — an anode and a cathode. The cathode is connected to the positive (+) connection on the battery and holds positively charged ions, and the anode is connected to the negative (-) connection and holds (you guessed it) negatively charged ions.
Between the two electrodes is what's called an electrolyte. The electrolyte in a lithium battery is (usually) an organic solvent paste that has a very large number of metallic salts (in most cases, that metal is lithium) as part of its makeup. This makes it electrically conductive — electricity can pass through it. The anode and the cathode are in the electrolyte and separated by a physical barrier so they can't touch.
When you discharge the battery (when you're using your phone and not charging it) the cathode pushes its positively charged ions away and the negatively charged anode attracts them. Electricity flows out from the anode, through your device, then back to the cathode. Yes, electricity travels through a loop and isn't "used up" by the thing being powered. When you charge your phone, the reverse happens and ions travel from the cathode through the electrolyte to the anode.
Lithium is the perfect element for rechargeable batteries: It's lightweight, easy to recharge and holds a charge for a long time.
When these ions come in contact with the charged atoms in an electrode, an electrochemical reaction called oxidation-reduction (redox) frees the charged electrons to travel out through the battery contacts, which are connected to the electrodes. This continues to charge the lithium ions in the electrolyte until there aren't enough left that can hold a positive charge that's strong enough to move through the electrolyte paste, and your battery will no longer charge.
Get the latest news from Android Central, your trusted companion in the world of Android
Lithium is the lightest metal — number three on the periodic table. It's also very excitable, making it easy to create a powerful chemical reaction. This makes it a near-perfect metal to use in a portable rechargeable battery. It's lightweight, easy to recharge and continues to hold a charge for a long time.
What can make a battery explode?

For starters, let's define what explode means in this case. The electrolyte paste inside a lithium-ion battery is extremely volatile. It can (and will) react violently to other metals, and has a very low (180-degrees Celsius) melting point. Inside a sealed battery casing, the pressure generated can build up until the casing is ruptured, then rapidly escape. The pressure carries out extremely hot electrolyte fluids which can cause other things to catch fire. Some lithium batteries are vented with an escape hole so they won't rupture under pressure. When the battery casing ruptures and superheated liquid filled with melted metals is expelled under pressure, it causes an explosion.
There are two easy ways to make a lithium battery explode — heat, and physical damage. Let's look at both.
Overheating and overcharging
This is the most common reason a battery fails. Something goes wrong in the charging circuit and the input power continues to drive the chemical reaction. One place in the battery will eventually get too hot and, since it is still being charged, it can't cool down, causing what's known as thermal runaway.
In this case, the hot portion starts to generate its own heat, which makes other areas in the electrolyte overheat, and they cause more spots in the battery to overheat. The heat expands the electrolyte and creates steam, building pressure until the battery casing splits and forces out all the pressure and some very hot, very sticky (and very flammable once exposed to the air) gooey liquid.
A thermal runaway can happen fast enough that you don't even feel the heat before the battery fails.
When such a rupture happens, it can cause physical damage to the things that are close to it — when holding a phone, circuit boards and glass or plastic. These materials can also catch fire from the heat, which in turn makes the escaped electrolyte ignite and act like napalm — fire that sticks to things until it burns through them or burns itself out.
The thermal runaway process can happen very quickly, and things can go from "normal" to catastrophic failure inside the battery before the heat is even transferred through your phone to your hands. Thankfully, the hundreds of millions of lithium batteries produced every year have an extremely low (almost statistically insignificant) failure rate due to thermal runaway, partially because of safety measures (subscription required) like non-flammable additives to the electrolyte and coatings.
When your phone tells you that it's too hot and won't charge or run at full speed, it needs to cool down so thermal runaway doesn't happen. Listen to the little pop-up and let it cool.
Mechanical damage
Lithium batteries are designed to be lightweight, deliver high output, and be easy to charge. This means that the outside shell and the barrier(s) separating the electrodes are very thin and light, with most of the weight coming from the parts that can actually power your phone.
Because the partitions and case are thin, they're fairly easy to puncture or tear. If the structure of the battery itself is damaged in a way that makes the electrodes touch, a short circuit will happen. The instant electrical discharge is explosive, which can (and will) heat the electrolyte and create pressure to push it out through any ruptures in the battery case. It's hot, it's flammable and it's in contact with a spark. That's a recipe for disaster.
It's hot, it's flammable and it's in contact with a spark. That's a recipe for disaster.
A thin casing is also a safety precaution, though it sounds crazy. Thinner metal is easier to rupture, so less pressure can generate inside a sealed case — essentially creating a vent hole. Pushing out flammable hot liquid under pressure isn't a good thing. Letting more pressure build until it ruptures a thicker case is worse.
Other metals coming in contact with the electrolyte paste can also create a spark that leads to failure. I'll let you search YouTube yourself to see incredibly silly people puncturing phone batteries to make them explode. The reaction to foreign metal does the same thing as a short, but on a smaller scale.
What about the Note 7?
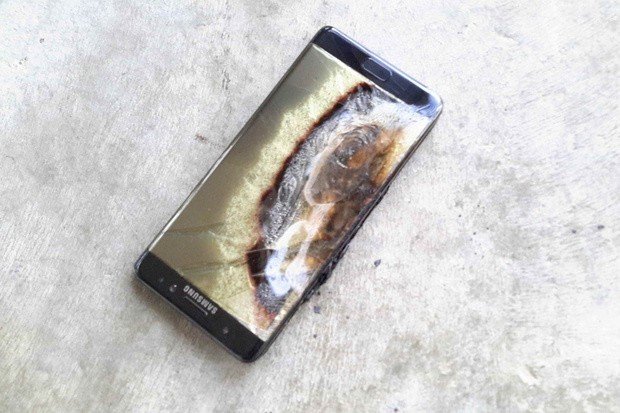
For starters, nobody but Samsung knows why batteries in some Note 7s have exploded. They sent out a short statement through their U.K, division that doesn't really make sense. The wording "the anode-to-cathode came into contact" sounds like they are describing a short, but as written it means nothing. I won't try to interpret something that isn't clear in a case like this. But I can say what I think as an armchair quarterback who has never seen a Note 7 explode or inspected one that has exploded on his own, a situation where the anode shorts to the cathode does make the most sense.
Because people have reported the Note 7 battery failing without it being plugged in and large portions of the phones we see in pictures are not burned, I'm going to assume that it's not a thermal runaway situation, even though Health Canada expicitly says the lone failure in Canada was from overheating,
Anyone outside of Samsung can only make an educated guess about what's happening.
A thermal runaway situation isn't as "instant" as a direct short, and the excited electrolyte solution would take a longer time to exit a burst battery then continue after an explosion ruptured the case. More than just a portion of the phone would be burned, as well as other objects in the immediate area. A heat explosion also would cause the battery to swell before the flexible casing would rupture, and with no room for expansion, the swollen battery would crack the outer shell of the phone. There are videos on YouTube that show how this happens, and here is a great (albeit old) example of someone circumventing the safety measures to make it happen. Someone would have mentioned the phone swelling up before it blew, I would think.
I also don't think any foreign objects or manufacturing leftovers are coming in contact with the electrolyte. I know this is a popular theory, but if a batch of batteries (we're not even sure how big a "batch" is in this case) all have small particles in the electrolyte, we'd be seeing a lot more Note 7s exploding.
A short inside the battery or in the charging circuit combined with an electrolyte that's been treated with non-flammable additives seems more likely to me. A quick explosion that releases a small amount of pressure and fluid at once could quickly burn itself out if nothing were in contact with the phone. When the phone is in contact with something that is flammable, say the seat of a Jeep, it could cause a fire.
Of course, there could be plenty of other factors that we don't know about. The manufacturing process used to create a lithium-ion battery for a phone makes them very safe to use. But there is also a lot that could go wrong.
A preliminary report by Samsung to Korean regulators blamed a production error that brought plates within the battery into contact, triggering "excessive heat." The report noted that more analysis was needed to pinpoint the exact cause.
Initial conclusions indicate an error in production that placed pressure on plates contained within battery cells. That in turn brought negative and positive poles into contact, triggering excessive heat. Samsung however stressed that it needed to carry out a more thorough analysis to determine "the exact cause" of battery damage.
What we do know about the Note 7 and its battery

We have no idea exactly what's happening with the Note 7 nor does anyone outside of Samsung. But we can make an educated guess based on the little evidence we're privy to. And in the end, it doesn't matter. The Note 7 has been determined by Samsung to be faulty and we're all supposed to stop using them and return them. A replacement without the issue is in production and already on its way to the sales channel. We don't need to know why it explodes to know that we should get rid of it.
Samsung recommends you power down your Note 7 and stop using it.
Samsung and your carrier (or other retailers like Target or Best Buy) have policies and methods in place that allow you to return your Note 7 without losing any money. Some offer loaner devices you can return when corrected Note 7 phones make their way onto the shelves, while others simply offer an outright refund. Either option is better than risking your hand or your face if the battery does fail.
We've compiled everything you need to know in one place, and we're also happy to answer any questions we can in the comments or on social media.

Jerry is an amateur woodworker and struggling shade tree mechanic. There's nothing he can't take apart, but many things he can't reassemble. You'll find him writing and speaking his loud opinion on Android Central and occasionally on Threads.
