Withings announces new ScanWatch with AFib and sleep apnea detection

What you need to know
- A 30-second ECG reading can be taken via the three built-in electrodes on the watch.
- An Sp02 sensor on the watch will track your oxygen saturation levels while you sleep to help detect sleep apnea.
- The new ScanWatch is due out in Q2 2020, pending CE and FDA clearance.
Withings, known for its connected health products and analog-style smartwatches, is back with a new watch that merges those two product lines even further. Today at CES, Withings announced the ScanWatch — a watch with Withings' familiar watch face but with more health tracking capabilities.
The ScanWatch's outside keeps the classic analog watch hands that we have come to know Withings for, but scales up the size of its POLED digital screen. The other change to the look of the watch is the inclusion of a new digital crown. This can be used for navigation on the watch and for selecting various functions, such as dismissing alarms, notifications, starting an ECG reading, and more.
ScanWatch is our most ambitious medical tracker and purposely designed to detect the early presence of AFib and sleep apnea - two related issues that are extremely common yet largely undiagnosed, despite their known impact on multiple health conditions," said Mathieu Letombe, CEO of Withings. "Worn throughout the day and night, ScanWatch provides an early warning system, collecting and sharing critical health data with users and their physicians.
Yes, that's right, the new ScanWatch has the ability to allow for an ECG reading directly on the watch — all in about 30 seconds. This is done using the three built-in sensors within the watch, two on the sides and one on the bottom. It's continuously monitoring your heart rate and looking for irregularities that could indicate AFib. If the watch senses there is something amiss in your heartbeat, it will prompt you to take an ECG reading.
The other new health tracking feature that comes to the ScanWatch is sleep apnea detection. Utilizing a Sp02 sensor, the ScanWatch can monitor your blood oxygen saturation levels to help detect if you may be suffering from inadequate breathing while you sleep. The watch also tracks your quality of sleep and includes Smart Wake-up to help wake you at the optimal point of your sleep cycle.
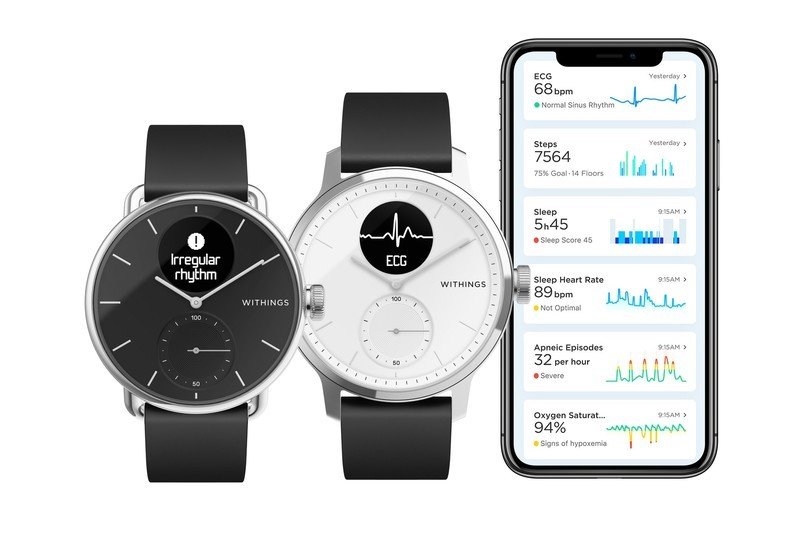
The health monitoring is saved to the Withings Health Mate app to review, as well as share with your doctor. The ScanWatch can also automatically track over 30 different exercises, your V02 Max levels, and integrate with over 100 third-party apps. Topping all of this off is 30-day battery life.
Product release is planned for Q2 2020, pending CE and FDA clearance, in the U.S. and Europe. Pricing will be €249/£229/$249 for the 38mm size and €299/£279/$299 to get the larger 42mm ScanWatch.
Get the latest news from Android Central, your trusted companion in the world of Android


